
People with darker skin tones may experience pigmentation changes with certain methods. If you do have angiomas removed with any of these methods, scarring is possible but uncommon. Cosmetic procedures may cause slight bruising. neodymium-doped yttrium aluminum garnet (Nd:YAG) laserĪ 2020 review found that people tended to prefer the PDL laser method over others.ĭepending on how many angiomas you have, you may need between one and three treatment sessions.potassium-titanyl-phosphate (KTP) laser.They use concentrated lasers that give off enough heat to destroy the lesion. Laser treatmentĭoctors can also use laser therapies to remove cherry angiomas. The number of treatments can depend on the size of the angioma. CryosurgeryĬryosurgery is a procedure that involves freezing off the angioma with liquid nitrogen. Then the dermatologist typically uses electrocauterization afterward to control bleeding. This procedure involves performing a shave biopsy to remove the angioma from the top portion of the skin. Electrocauterizationĭermatologists can remove smaller angiomas with electrocauterization, which uses an electric current delivered by a tiny probe.įor this procedure, you’ll also have a grounding pad placed somewhere on your body to ground the rest of your body from a surge of electricity. If you use this eHealthMe study on publication, please acknowledge it with a citation: study title, URL, accessed date.While cherry angiomas typically do not require treatment, dermatologists can remove them for cosmetic reasons.Ī doctor may also recommend removal if you have one in an area that’s easily bumped, which can lead to regular bleeding.Ī few common procedures can remove angiomas. 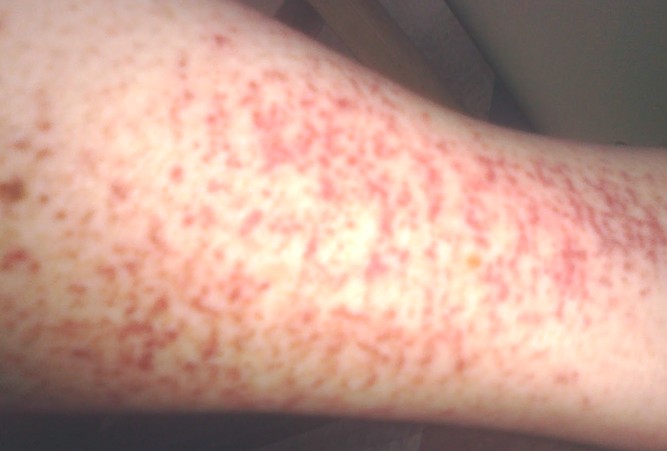
The use of the eHealthMe site and its content is at your own risk. Every effort has been made to ensure that all information is accurate, up-to-date, and complete, but no guarantee is made to that effect. Different individuals may respond to medication in different ways. Our phase IV clinical studies alone cannot establish cause-effect relationship. WARNING: Please DO NOT STOP MEDICATIONS without first consulting a physician since doing so could be hazardous to your health.ĭISCLAIMER: All material available on is for informational purposes only, and is not a substitute for medical advice, diagnosis, or treatment provided by a qualified healthcare provider. Our analysis results are available to researchers, health care professionals, patients ( testimonials), and software developers ( open API). Results of our real-world drug study have been referenced on 700+ medical publications, including The Lancet, Mayo Clinic Proceedings, and Nature. We study millions of patients and 5,000 more each day. With real-world medical big data and proven AI/ML algorithms, eHealthMe provides a platform for everyone to run phase IV clinical trials. You can discuss the study with your doctor, to ensure that all drug risks and benefits are fully discussed and understood. The FDA has started to use real-world data to monitor postmarket safety and adverse events, and payers to support value-based payment program. The 21st Century Cures Act, passed in 2016, places additional focus on the use of real-world data to support regulatory decision making. These programs potentially contribute to the variability of the quantity and quality of evidence supporting recent drug approvals.Īll these suggest an ongoing need for continued evaluation of therapeutic safety and efficacy after drug approvals. When the drugs are approved and released in the market to a much bigger and ever-changing public, it is not uncommon for drugs to behave differently in the real world than in clinical trials.ĭuring the past 30 years, special regulatory programs are available to the FDA including Fast Track (1988, in statute 1997), Priority Review (1992), Accelerated Approval (1992), and Breakthrough Therapy designation (2012). 
The guidelines ensure that only a selected set of people are chosen due to time and costs. All clinical trials have guidelines about who can participate. New drugs have to go through a series of clinical trials before they are approved by the FDA.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
